10 Sep What do you need to know about Thermal Energy?
Thermal Energy Transfer / Greenhouse Effect
Some Background
IB calls this topic Thermal Energy Transfer, but really it’s about the greenhouse effect and climate change. Calling it Thermal Energy Transfer it sounds more physics-y.
Climate change is of course a controversial subject. Before I started teaching physics, I wasn’t quite sure what to believe about it. But now that I had learn it well enough to teach it, I understand that the basic physics of the greenhouse effect are irrefutable, and that there is no way that adding CO2 to the atmosphere could NOT have a warming effect.
We still can’t predict with high confidence exactly what the effects will be, but the direction is clear: the earth has to get warmer. It has to. It doesn’t matter whether you think the temperature records of the last few decades demonstrate this or not. Even if you think the climate hasn’t changed yet, just wait a while longer: it’s going to be warmer in the future than it would otherwise be if we did not pour CO2 into the atmosphere. Guaranteed. We have raised the CO2 concentration very fast, but it takes time to warm up something as big as the earth, so the effects of what we’ve already done won’t show their full force for
How much warmer will it get, and how fast? This is hard to tell. At least a few degrees, and certainly at a rate that is much faster than what the earth has previously experienced from natural causes. What should we do about it? That’s a question that goes beyond science to economics, politics, and even morality.
Here’s an excellent video by Veritasium that addresses the most popular misconceptions about climate change:
On to the physics…
A major component of this topic is understanding “Black Body Radiation“. Every object emits electromagnetic radiation. The temperature of the object determines how much power is radiated, and how that power is distributed across the frequency spectrum. It depends on the color of the object, but a perfectly black object will give you the most radiation for any particular temperature. Perhaps surprisingly, a lot of objects behave almost like black bodies, including the earth and the sun, so we will use the black-body model for them.
We have complicated equations that can tell us exactly how much radiation there will be at any particular wavelength or frequency for any temperature of the object. But we don’t need that level of detail to understand what’s going on, so , we are going to focus on just two ways of describing the power radiated by a black body:
- The amount of power radiated goes up very fast as temperature goes up, and it gets much smaller as temperature goes down. The Stefan-Boltzmann equation quantifies that relationship.
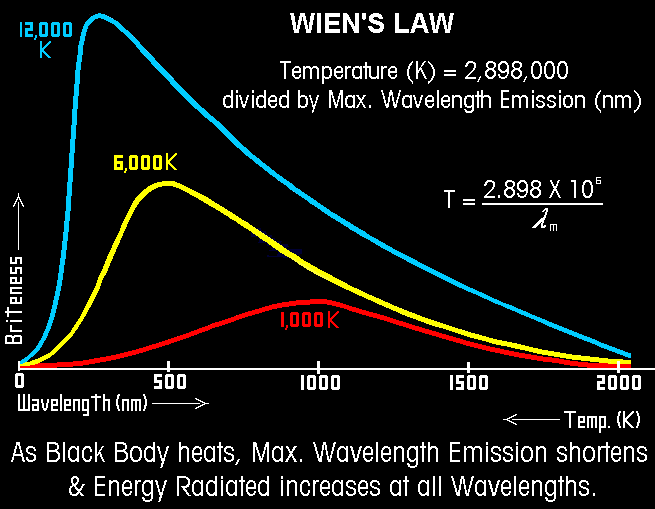
- The wavelength distribution of the radiation changes with temperature. Cooler temperatures push the peak toward infrared. Infrared waves are lower energy, lower frequency, longer wavelength. Hotter temperatures push the peak toward ultraviolet. UV rays are higher energy, higher frequency, shorter wavelength. So it makes sense that the peak shifts toward UV as the object gets hotter. Wien’s Law tells us how to calculate exactly where the peak is on the frequency spectrum.
You can look at how the distribution of the radiation changes with temperature with this excellent simulation of the black-body effect:
https://phet.colorado.edu/sims/blackbody-spectrum/blackbody-spectrum_en.html
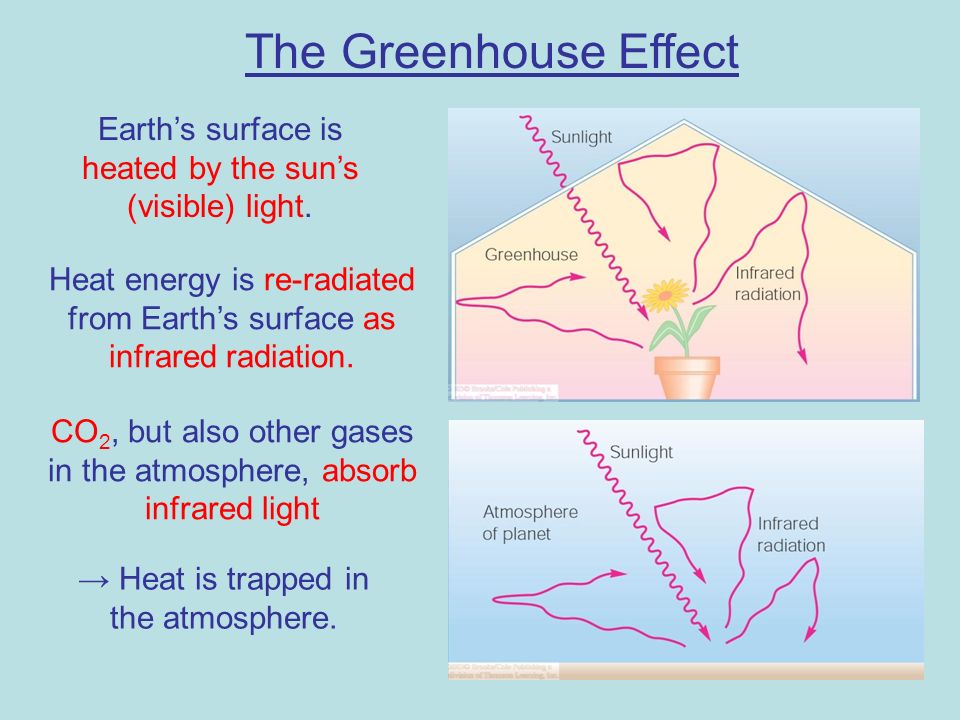
Wien’s Law explains why the sun radiates all across the visible spectrum: the sun’s surface is very hot, about 5000-6000 Kelvin. Wien’s Law also explains why the earth radiates only in the infrared part of the spectrum: the earth’s temperature, 288 K, is very cool compared to the sun. The reason this is important for this topic is that the earth’s atmosphere only absorbs infrared radiation, not visible or ultraviolet light. This means that most of the sun’s light that does not reflect off the atmosphere will pass all the way through the atmosphere and be absorbed by the earth’s surface. It also means that when the earth re-radiates that energy, the radiation will all be in the part of of the spectrum that the atmosphere does absorb. This is the greenhouse effect: light energy comes in easily but can’t get out so easily. We need it, or else the planet would be far too cold for us (about 255 K)!
Stefan-Boltzman tells us what temperature the atmosphere and the earth’s surface have to reach in order for the earth to find an equilibrium where the energy coming into each of them is equal to the energy going out. That’s when the temperature of each one will be stable. If more energy is coming into an object than going out, the object’s temperature will rise. Stefan-Boltzmann tells us that rise will cause an increase the power going out, until the energy flows balance again.
When the atmosphere absorbs infrared radiation coming up from the earth’s surface, the atmosphere heats up and radiates more, too. It radiates in all directions, so half of this atmospheric radiation goes out to space, but half of it goes back down toward earth’s surface. So now earth’s surface has TWO sources of incoming radiation: the original solar radiation, AND the infrared radiation coming down from the atmosphere. The earth’s surface has to heat up to restore the balance. This is the earth’s version of the greenhouse effect: the absorption and re-radiation of infrared energy by the atmosphere.
Why does the atmosphere absorb infrared? It’s because the atmosphere has gas molecules in it that are very good at absorbing infrared. These include water vapor, CO2, methane, and nitrous oxide. These come from natural and man-made sources. The reason these molecules are good at absorbing infrared has to do with the laws of quantum mechanics, but we can understand it in a simplified way by considering that molecules are atoms connected by chemical bonds, and these atoms behave a little bit like masses connected by springs. Mass/spring systems have resonant frequencies, and the resonant frequencies for the greenhouse gas molecules match the frequency of infrared light. This makes them good at absorbing photons of light of that frequency, and then re-emitting that energy at the same frequency. It’s similar to the way a swing will absorb your energy if you pump your legs with the right timing, but you won’t go anywhere if you stick your legs out at the wrong frequency, too rapidly or too slowly.
Light that does not match the resonant frequency of the molecules will not be absorbed and will pass right through. This is why most of the sunlight reaches the ground but the infrared radiation gets trapped on its way out.
Enhanced greenhouse effect
The greenhouse effect is natural, and we need it to keep the planet warm enough to be habitable. But by burning fossil fuels, humans are adding large quantities of CO2 into the atmosphere, and this creates an enhanced greenhouse effect, which must result in planet that is warmer than it would otherwise be. I say “must” because it is just a matter of physics. We KNOW that earth radiates infrared and why it does that; we KNOW that burning fossil fuels adds CO2 to the atmosphere, and why it does that; we KNOW that CO2 absorbs infrared, and why it does that; we KNOW that when the atmosphere absorbs more infrared the earth has to get hotter, and why that’s the case. All of this is demonstrable in a laboratory!
If we are adding lots of CO2 to the atmosphere, we should be able to measure that, and we can. We know that the concentration of CO2 in the atmosphere is now over 400 parts per million. The norm over the last 800,000 years has been more like 200 or 250. NEVER has there been this much CO2 in the atmosphere in the last 800,000 years, not even close. And the amount has never risen at the rate that it has risen over the past 70 years or so. The earth has “normal” cycles of increases and decreases in CO2, but those occur over tens of thousands of years, not decades.
We know that the CO2 in the atmosphere came from burning fossil fuels because it has the right chemical “signature” (the right blend of isotopes). We know how much fossil fuel humanity has burned, and that matches up with the increase of CO2 in the atmosphere and oceans.
We also know that over the last 800,000 years, when CO2 has increased, so has the temperature. Skeptics like to suggest that maybe the temperature causes the CO2 to go up rather than the other way around, and it’s true of course that correlation never proves causality. But how could more CO2 not cause the temperature to rise? We know the physics! When we understand the mechanism of a causal relationship AND we see correlation, then it is silly to continue to pretend the causality might go the other way.
What happens in these cycles is that an initial warming starts a release of previously trapped CO2, and then that released CO2 causes additional warming, in a feedback loop. In past cycles, there were obviously non-human causes of the initial warming that released CO2. Most commonly these causes were astronomical, having to do with wobbles in the way the earth orbits the sun. This time, the cause is human emissions of CO2. Either way, we know what happens next: feedback creates additional warming. That’s what’s happening now.
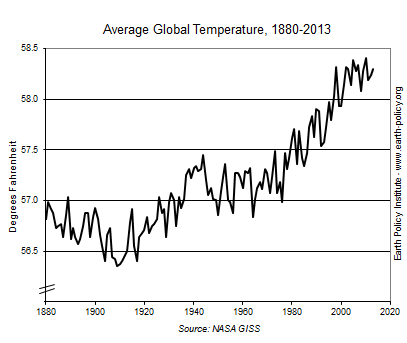
Here’s a graph of earth’s temperature more recently:
There are other greenhouse gases besides CO2. Methane is a much more potent absorber of infrared, but it breaks down in the atmosphere, while CO2 does not. Water vapor is the most significant greenhouse gas, but when there’s too much water in the air, it rains, and vapor is removed. But, as the planet warms, the air holds more water vapor, and this additional vapor is an important source of warming – it roughly doubles the warming effect of our CO2 emissions.
The history of earth’s climate
You might wonder how we could possibly know the CO2 concentration and temperature over a period of 800,000 years. An important source of information about the history of the climate is buried in Antarctic ice. Every winter, snow falls in Antarctica, and because it is so cold, it never melts. As more snow falls on top of the older snow, the older snow becomes compressed into ice, and each winter’s snow is recorded as a distinct layer of ice, just as each summer is recorded in the growth rings in a tree’s trunk. If we dig into the ice, we can count the layers, and we can figure out how old the ice is that way.
Trapped inside each layer of ice are tiny air bubbles. Eight hundred thousand layers down, there’s air from 800,000 years ago, like an air fossil! We can extract that air and measure the CO2. We can also figure out what the temperature was when the air was trapped by comparing the presence of certain oxygen isotopes in the air.
There are other sources of variation of CO2 and temperature besides human activity, of course. Small variations in the path of earth’s orbit around the sun produce cyclical variations that are clearly evident in the 800,000 year record. These cycles have a period of about 100,000 years and are responsible for the arrival and end of ice ages. As the earth emerges from an ice age, CO2 is released from the oceans, and then this CO2 amplifies the warming effect until the earth’s orbit takes it back into a phase of cooling. These cycles are far too slow to explain any of the CO2 changes or temperature changes we have seen in the past 100 years. Other sources of temperature variation include volcanic activity and solar activity. Volcanoes emit much, much less CO2 than humans produce by burning fossil fuels, and solar activity does not track the changes we see happening on earth.
What else IB wants you to know
You should be able to figure out what the intensity of the sun’s radiation is at earth’s distance from the sun. It’s about 1400 W/m^2, and it’s called the solar constant, S.
You should also be able to explain how 1400 W/m^2 absorbed by a circle whose radius is equal to that of earth becomes 350 W/m^2, or S/4, when it gets spread over the spherical surface of the earth.
You need to know the definitions of albedo and emissivity.








Sorry, the comment form is closed at this time.